A New Clinical Research Study for People with Chronic Cough
About This Study
Aldeyra Therapeutics is sponsoring a medical research study (also called a clinical trial) to learn more about an experimental drug called ADX-629 for adults with chronic cough.
Clinical trials help doctors and scientists learn about potential new drugs by either comparing it to a placebo or an already approved medication. All eligible participants in this study will receive ADX-629 at some point.

You may be eligible for this clinical trial if:
You are 18 to 80 years old
Have had a cough for over a year
Are a non-smoker
You are willing to attend 10 clinic visits in the next 3 months
Participating in a clinical trial may involve unforeseeable risks and does not guarantee that your symptoms improve.
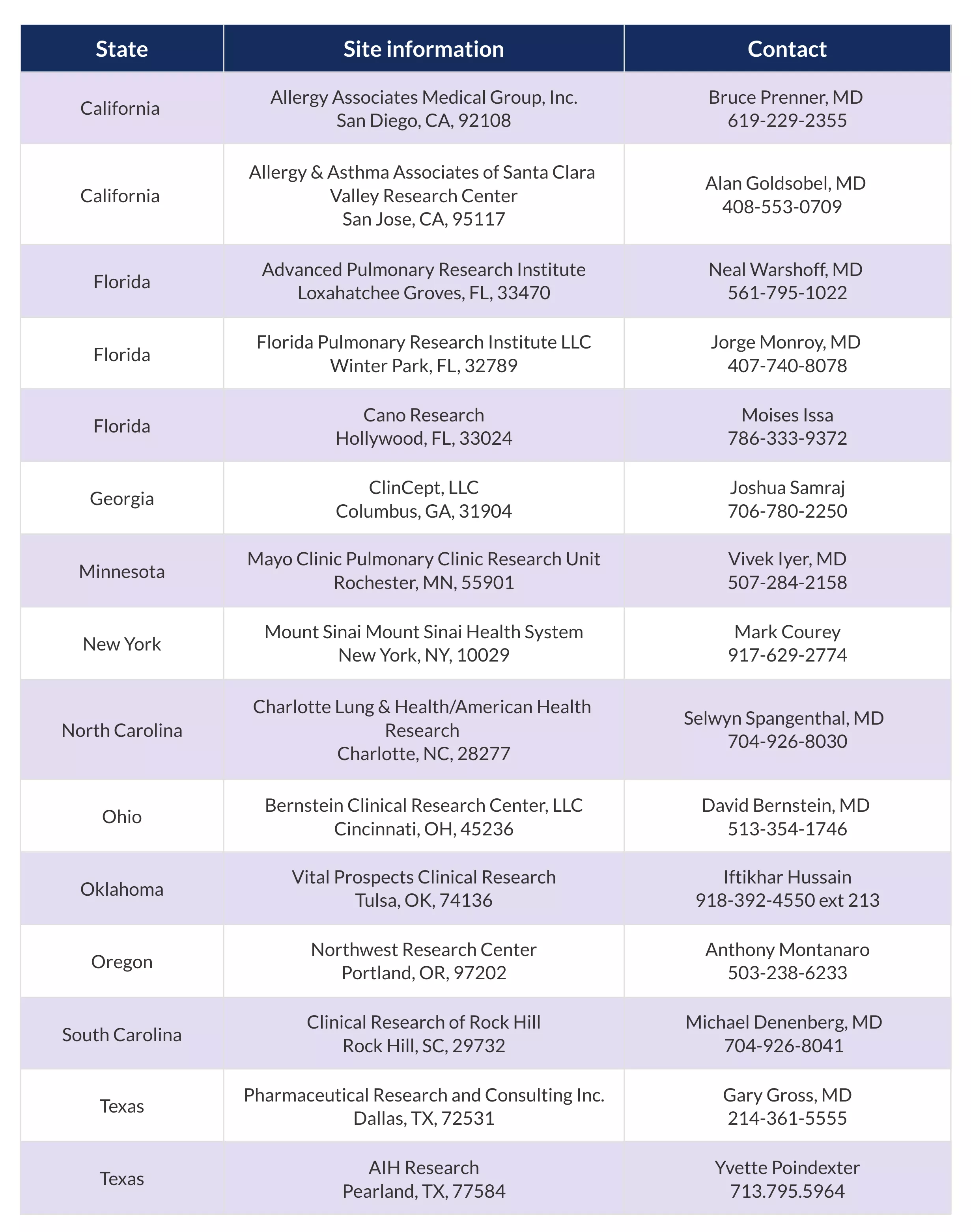
A full description of this trial is available on https://www.clinicaltrials.gov with the ClinicalTrials.gov Identifier: NCT05392192

Eligible trial participants will:
- Be connected with a trial doctor at a research clinic nearest them
- Receive study information and learn more about their cough
- Undergo health assessments and study procedures
- Receive study drug (ADX-629) at no cost to them
- Be compensated (up to $2,500) for completing all clinic visits
- Support research and the development of potential new drugs
About Chronic Cough
Chronic cough is a cough that lasts 8 weeks or more and may be caused by other health conditions, including asthma, allergies, or gastroesophageal reflux (GERD). There are over-the-counter and prescription cough medications available to help with cough. (Click here to learn more about chronic cough.)
If you have tried medication and still have a cough that will not go away, you may be eligible for a clinical trial sponsored by Aldeyra Therapeutics.

Did You Know?
All prescription drugs that are taken today were once studied in clinical trials. Scientists cannot study new, ground-breaking medications without the help of volunteers. New medications cannot be prescribed unless they are found to be safe and effective in clinical trials.

A large number of experimental drugs never get a chance to be researched because scientists are unable to find enough volunteers for the clinical trial.

Any qualified, experienced medical doctor can become a trial doctor even your family physician or pulmonologist.

Trial sites can be any hospital, medical clinic, or doctor’s office where trial-related assessments take place.

You do not have to pay or have health insurance to sign up for a clinical trial. Volunteers often recieve compensation for participating in a clinical trial.
Frequently Asked Questions
What is a research study?
Medical research studies (or clinical trials) help doctors and scientists learn about experimental drugs, including how well they work and how safe they are. Clinical trials may study the experimental drug (or study drug) by itself, compare it to an approved medication, or placebo.
What should I expect if I join a clinical trial?
Every clinical trial is different, so it is important to always read all the information that is provided by the trial doctor and research staff. The trial participants will always be informed of the procedures that they can expect to undergo before they decide to join the clinical trial. Participation in a clinical trial is usually broken down into three separate stages: Screening, Study treatment, and Follow-up.
- Screening: The purpose of the screening period is to make sure participants are suitable to take part in the trial.
- Treatment: If a participant is deemed eligible, they will officially be enrolled in the clinical trial and receive the study drug.
- Follow-up: Participants will complete one last visit after they are done with the study drug.
What assessments will be performed?
There are common assessments that are performed during most clinical trials. Here are some examples of trial activities you can expect:
- Answer personal questions about your medical history and current health conditions
- Provide a list of your current medications
- Get your blood pressure and heart rate measurements taken
- Have a physical exam performed by the trial doctor
- Undergo blood draws for routine laboratory testing
- Receive a electrocardiogram (ECG) to measure your heart’s activity
- Take the study medication as directed and return it to the clinic when done
- Tell the trial doctor of any changes in your health or changes to medications
Remember participating in a clinical trial is always voluntary. Even after signing up, you can leave at any point during the trial.
How long will this trial last?
Every clinical trial is different. Volunteers can participate clinical trials anywhere from a few days to a few years. The trial doctor will always tell you how long the trial is before you decide to join.
Do I have to pay to join a clinical trial?
You do not have to pay or have health insurance to sign up for a clinical trial. The trial drug and trial-related procedures are provided to participants at no charge to them or their health insurance company. Clinical trials often provide participants compensation (or a stipend) for their time and effort.
How do I sign up?
There are many different resources patients can use to find clinical trials. Your doctor can also help you find clinical trials that you may qualify for.
Responsum Health has collaborated with Aldeyra Therapeutics to help raise awareness and connect patients to a new clinical trial for chronic cough! Find out if you’re eligible to participate in a Chronic Cough Study here.
Ready to learn if you can take part in this research study?
Additional Information and Resources
Information about clinical trials and how to find ones in your area.
- Mayo Clinic:
- About Clinical Studies: https://www.mayo.edu/research/clinical-trials/about-clinical-studies
- The Clinical Trial Journey: https://www.youtube.com/watch?v=iWqQiJeP5ac&ab_channel=MayoClinic
- Research Match: https://www.researchmatch.org/
- National Insititute of Health: https://www.nih.gov/health-information/nih-clinical-research-trials-you
- CenterWatch Trial Finder: https://www.centerwatch.com/clinical-trials/